CRISPR Used to Treat Fatal Muscle Disorder

Image Courtesy: Technology Networks
CRISPR-Cas9, the revolutionary gene-editing technique, has been used on blood samples for treating Duchenne muscular dystrophy (DMD), a rare and fatal muscle disease. More than 3,000 genetic mutations are responsible for the disease and using CRISPR, scientists were able to correct these mutations.
The research was carried out by a team of scientists based in US and Germany, and their results were published in Science Advances last week. Eric Olsen, the corresponding author of the study, said, “This disease has defied every therapy that’s been tested so far. It’s a really tough one.”
DMD afflicts one out of every 5,000 males. It causes muscle weakening all over the body, often resulting in teenage deaths from weakened heart muscles. It is generally seen in boys with its effects beginning from infancy.
DMD causing mutations result in a failure of the production of the protein dystrophin. Dystrophin is fundamental in improving the strength of muscle fibres and its absence causes degeneration of muscle cells. The leading cause of death from DMD is a condition called dilated cardiomyopathy (DCM). The hearts of people suffering from DCM are unable to pump blood effectively. There is no treatment which can cure any of the conditions caused by DMD thus far.
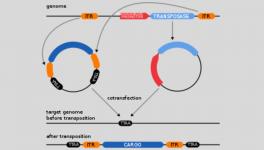
Performing gene therapy on DMD mutations posed significant challenges. “DMD gene is the largest known gene in the human genome, encompassing 2.6 million base pairs,” the authors write in the study. Because of its large size and complex structure, the rate of mutations is high. Correction of these thousands of mutations using a targeted technology such as CRISPR then could not have been easy.
What made it possible is the fact that DMD mutations in humans occur in clusters termed as “hotspots”. While each and every individual mutation could not be targeted, 60% of them could because of the existence of these hotspots.
CRISPR was used to successfully target areas within and around the mutated genes and replace them with healthy ones. After treatment, production of dystrophin began again in muscle cells. Tests of this technique were conducted on live mice as well as dogs prior to experiments on human cells in a petri dish. The mice have been observed for an year now and have remained healthy while continuing to produce dystrophin.
One of the major concerns of using CRISPR is that the treatment can go off target and cause unwanted mutations, resulting in other problems. But no off-target edits seem to have been observed in this study. Olson says follow up studies are needed to confirm this.
Genetic mutations result in various disorders for which no easy treatment is available. With the increase in experimental success of CRISPR, hope for one day treating these disorders is also increasing. Clinical trials using CRISPR on humans have only be approved in US and China till now. However, ethical challenges of using this technology for purposes other than treatment of illnesses remain unaddressed.
Olson said clinical trials could start within a couple of years if studies continue smoothly.
Get the latest reports & analysis with people's perspective on Protests, movements & deep analytical videos, discussions of the current affairs in your Telegram app. Subscribe to NewsClick's Telegram channel & get Real-Time updates on stories, as they get published on our website.