Blue is a Difficult Colour!

The entire sky might appear blue, the oceans might appear blue, but throughout history, making a blue pigment has been a hard nut to crack. And not only human-made blue pigment is rare; blues are also scarce in nature. Humans started making pigments at least 1,00,000 years ago, from red and yellow ochre, but couldn’t achieve a blue pigment. Lapis lazuli, the brilliant blue coloured metamorphic rock has been used as a semi-precious stone since antiquity. The Babylonians and the Egyptians used bits of Lapis lazuli in their statues and frescoes as colouring material. However, making a pigment out of it was a laborious task; lapis lazuli had to be ground to powder to get the ultramarine pigment which could then be used for painting. This method of obtaining a blue pigment was only discovered in the sixth century B.C.E.
Recent evidences from the burial site of Catal Huyuk in Turkey revealed that people used to grind azurite, the blue mineral, to fine powder around 9,000 years ago. Azurite, as the findings suggest, was also used in cosmetics during those days.
Quest for Synthetic Blue Continues Since Ancient Till Today
The first ever synthetic blue pigment came from the Egyptians—the ‘Egyptian blue’. They mixed sand, plant ash and copper to create it some 5,000 years ago. The following timeline appearing in Science would surely give a hint to understand the hunt for blues:
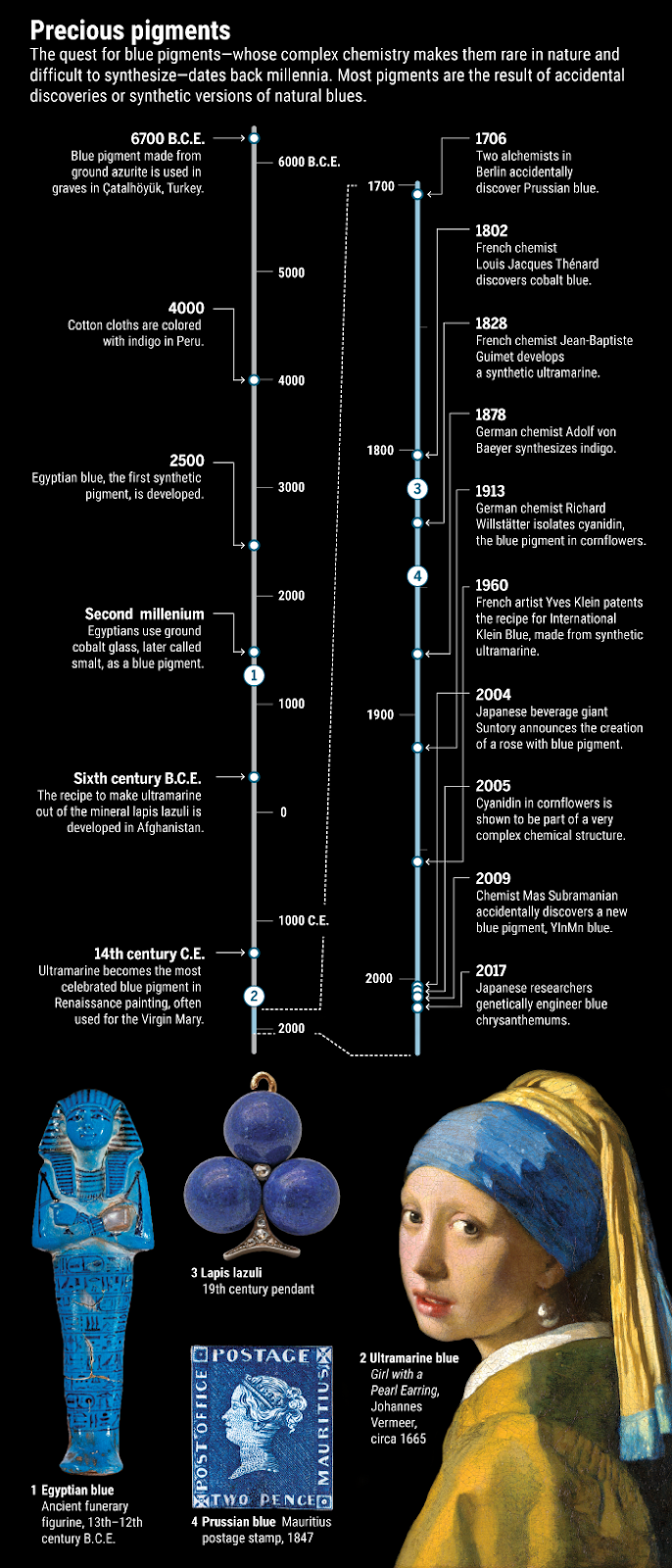
Travelling down to the nineteenth century, chemists raced to create a synthetic ultramarine. Significant efforts were also made to synthesise indigo, the deep blue dye extracted from plants. BASF, the chemical giant based in Germany, invested some 18 million gold marks for the synthesis of indigo. This amount accounted for more than the worth of the company at that time. These blues became an object for extensive search for the booming chemical industry.
Indigo has had a long history in India too, with the Indigo movement being one of the first organised struggles against the British colonial regime in the country.
Indigo Revolt in India
The Indigo Revolt (Neel Bidroh) which took place in Bengal in 1859-60, was a revolt by the indigo farmers against British planters. Indigo as a dye used to be very popular in Europe those days and the British formulated a policy of indigo farming that favoured the British traders over the farmers. The farmers were forced to sell indigo at a non-profitable rate so as to maximise the profit of the British planters.
Indigo farmers revolted in Nadia district of Bengal where they refused to grow indigo any more. The revolt met with brutality by police. Finally by the end of 1860, Indigo farming was literally washed away from Bengal.
Blue Flowers and the Accidental Discovery of Inorganic Blue
Even in the 21st century, scientists continue with the age-old quest for new blues. In 2004, researchers from Japan reported to have developed the world’s first blue rose. But right after, the concern arouse that it was not too blue. Yoshikazu Tanaka, the lead scientist behind the discovery also agreed that it could have been bluer. But Tanaka, untired even after 15 years, is trying his hand in bringing out blue in cut-flowers like chrysanthemums, carnations or tulips. None of these naturally occur in blue. Roses too have been bred over decades in various hues of yellow, pink and red, but never blue.
In 2009, Mas Subramanian of Oregon State University was trying hard to develop a multiferroic—a material that is a combination of magnetic and electronic properties. Once a student, while working in Subramanian’s lab, mixed some inorganic chemicals and it turned out that the mixture changed to a deep blue one. It was an accidental discovery of a mixture of inorganic compounds that can produce a deep blue material. Named as YInMn blue, this material made the headlines in US newspaper. The thing is blue is still very hard to make.
Why is Blue a Difficult Colour?
Any pigment shows its colour based on the principle of absorption and radiation of light wave. For a dye or pigment to appear blue, it needs to absorb red light. In doing so, photons boost electrons in the pigment from one energy level to the next. But red light is the one having the lowest energy in the visible spectra. So, in order to boost electron from one energy level to the next by red lights, the energy levels must lie very close together. These kinds of closely spaced energy levels are found only in molecules that are very complicated and hard for organisms to make.
In plants as well, we could see different pigments ranging from green (chlorophyll), orange (carotenoids), red, yellow, etc. But blue remains rare in this case as well. The only plant pigment that produces blue colour is anthocyanin.
The accidental blue that was discovered in Mas Subramanian’s lab was also due to complicated chemistry of the mixture. The blue colour is created by a manganese ion surrounded by five oxygen atoms and the structure resembles two pyramids glued together at their bases—a rare geometry seen in natural minerals.
Blue still remains a difficult colour.
Get the latest reports & analysis with people's perspective on Protests, movements & deep analytical videos, discussions of the current affairs in your Telegram app. Subscribe to NewsClick's Telegram channel & get Real-Time updates on stories, as they get published on our website.























