COVID-19: 41 Vaccine Candidates in Clinical Trials, 191 in All, Says WHO Report
Image Courtesy: The Financial Express
With the novel coronavirus not showing signs of stopping, the race for a vaccine is on with efforts underway in many countries and clinical trials on the up. According to the World Health Organisation’s ‘Draft landscape of COVID-19 candidate vaccines’, released on 30th September, as many as 191 candidates are in various stages of development.
Among these, 41 candidate vaccines have made their way into clinical trials. Out of the 41, ten candidates have entered the third phase of clinical trials. The third phase is the highest level for a clinical trial before a vaccine is usable by the public.
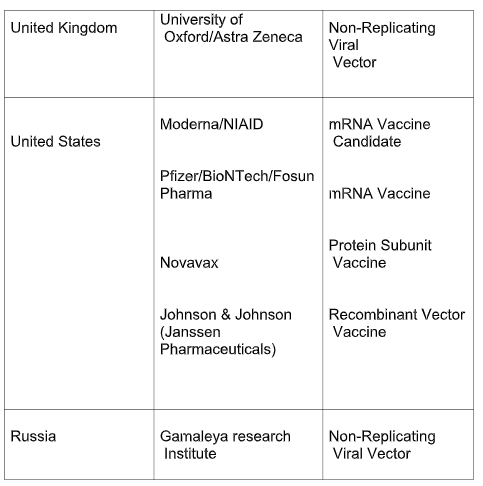
The table below lists the candidate vaccines that have entered phase III of clinical trial, along with their developers, country of origin and the type of vaccine being developed. The data has been taken from the WHO report.
The Indian Scenario:
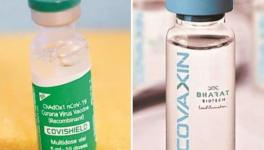
In India, three candidate vaccines are at different stages of clinical trials. India’s indigenous candidate, Covaxin, manufactured by Bharat Biotech in collaboration with the ICMR (Indian Council of Medical Research) and NIV (National Institute of Virology) is in phase I and phase II stages of clinical trials.
The vaccine candidate being developed by Oxford University has been named Covishield in India and is being produced by the Serum Institute of India (SII). ICMR and SII are now jointly undertaking phase II/III trials in the country – an observer-blind, randomised controlled trial. According to a report, Covishield phase II and phase III clinical trials are progressing smoothly in India. Its phase III trial began in Pune on September 17 and on September 23 in Mumbai.
Some participants reportedly have taken the second dose of the candidate vaccine and the trials show no major adverse effects as of now.
The third vaccine candidate under human trial is ZyCoV-D, developed by Zydus Cadila. This candidate is now under phase I and phase II of clinical trials.
ICMR has recently launched a web portal dedicated to vaccine, which can be found here. The ICMR vaccine portal has information about COVID-19 vaccine clinical trials being conducted in different parts of the country. It includes the clinical registry details of each individual vaccine candidate, which comprises the name and addresses of the hospitals or medical institutes where the clinical trials are being conducted.
Get the latest reports & analysis with people's perspective on Protests, movements & deep analytical videos, discussions of the current affairs in your Telegram app. Subscribe to NewsClick's Telegram channel & get Real-Time updates on stories, as they get published on our website.