COVID-19: 44 Vaccine Candidates Under Clinical Trial Across World

Image Courtesy: Bloomberg
While the COVID-19 pandemic continues its grip across the world, the race for a vaccine is on with efforts underway in many countries and several vaccines at the level of clinical trials. According to the “Draft Landscape of COVID-19 candidate vaccines” prepared by the World Health Organisation (WHO), till October 19, when the draft landscape was updated last, as many as 198 vaccine candidates aimed against COVID-19 are in various stages of development. Out of these, 44 candidates have entered the level of clinical trial, that is, applying on human subjects.
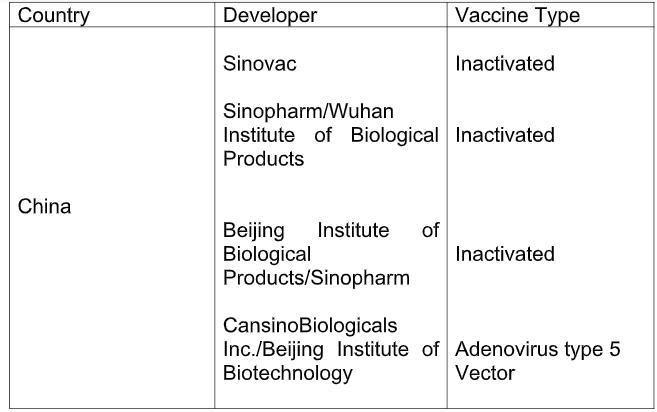
Again, out of the 44 candidates in clinical trials, those in the third stage are 10. Interestingly, as per the draft landscape updated till September 30, number of phase 3 candidate vaccines were also 10.
The table below lists the candidate vaccines that have entered phase III of clinical trial, along with their developers, country of origin and the type of vaccine being developed. The data has been taken from the WHO report.
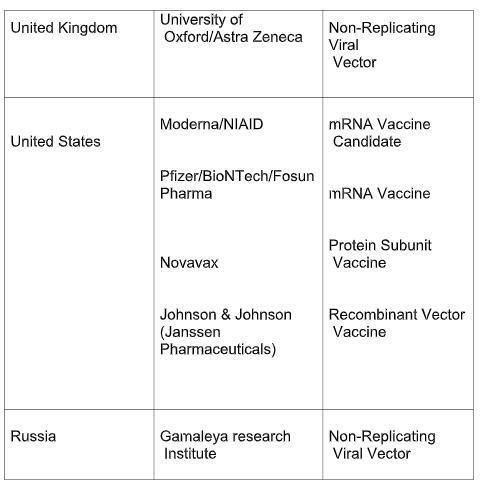
The speed at which these vaccine designing trials are proceeding is unprecedented. Although considerable resources and efforts have been mobilised for the development of vaccine against the novel coronavirus or SARS-CoV-2, the search for the vaccine has remained inconclusive so far. However, experts hope that a vaccine could be available anywhere between end of 2020 and middle of 2021. Till now, only two vaccine candidates have been approved, that too amid wide-scaled controversies and criticisms. Sputnik V, developed by Gamaleya Institute, Moscow and EpiVacCorona, also developed in Russia, are the two vaccines that have received the approval from Russian Government. Notably, neither of these two candidates completed phase 3 clinical trials before receiving the approval.
In India, three candidate vaccines are at different stages of clinical trials. Covaxin, which could be called India’s indigenous candidate and manufactured jointly by Bharat Biotech, ICMR (Indian Council of Medical Research) and NIV (National Institute of Virology) is under phase 2 clinical trial, according to ICMR’s vaccine portal.
Covishield, the vaccine candidate in India developed by Oxford University and produced by SII (Serum Institute of India) is under 2nd and 3rd phase of clinical trial.
And the third candidate is ZYCoV-D developed by Zydus Cadila which is undergoing phase 1 and 2 clinical trial.
Get the latest reports & analysis with people's perspective on Protests, movements & deep analytical videos, discussions of the current affairs in your Telegram app. Subscribe to NewsClick's Telegram channel & get Real-Time updates on stories, as they get published on our website.