Delhi High Court Averts Public Health Crisis, Sets Aside Oxytocin Ban
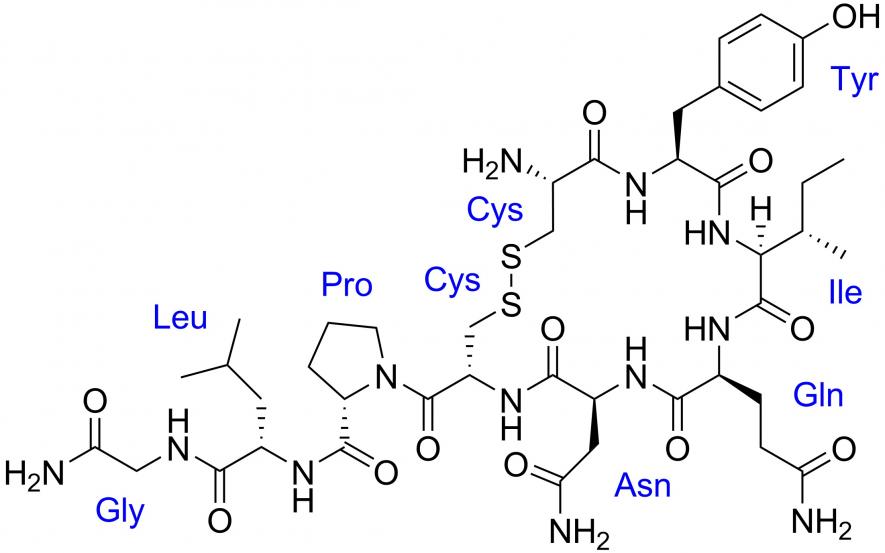
Chemical structure of oxytocin. | Image Courtesy: Wikipedia
NEW DELHI: Citing the Right to Life under Article 21, the Delhi High Court on December 14 quashed a notification by the central government that had endangered the lives of pregnant women in the country — and gave precedence to cattle instead — by banning the manufacture and distribution of life-saving Oxytocin drug and restricting its production to just one public-sector company that had no history of producing it.
Also Read: Oxytocin Ban: A Public Health Crisis in the Offing
Spurred by allegations that Oxytocin was being misused in the dairy sector to increase milk production by cattle and was impacting the animals’ long-term health, the Ministry of Health & Family Welfare on April 27, 2018 had issued a notification that prohibited all private companies from manufacturing Oxytocin for domestic use and stopped its distribution through retail.
The ban was initially supposed to come into effect from July 1, after which the date was extended to September 1, but it was followed by two stay orders of a month each by the Delhi high court — after the All India Drug Action Network (AIDAN) and three private companies producing the drug moved court.
The ministry had designated Karnataka Antibiotics & Pharmaceuticals Limited (KAPL) as the sole manufacturer of the drug — even though, as the judgement noted, “till mid 2017, KAPL had no manufacturing ability to produce the drug.” What’s more, “as per the submission of KAPL, the earliest production of Oxytocin can be possible only after 3 years,” the court noted
Oxytocin is given to pregnant women to prevent and treat postpartum haemorrhage (excess bleeding immediately after childbirth) — a leading cause of maternal mortality worldwide. It is also used to induce labour for the delivery of the child.
Also Read: India Records Decline in Maternal Mortality
In fact, Oxytocin is recognised as “essential” in India and is included in the National List of Essential Medicines, 2015 (NLEM), that is published under the First Schedule to the Drugs (Prices) Control Order, 2013. It is even included in the 20th World Health Organization (WHO) Model List of Essential Medicines, March, 2017.
Oxytocin is recommended by the World Health Organisation as the first line drug for prevention and treatment of postpartum haemorrhage — which causes approximately 69,000 maternal deaths worldwide every year, as the judgement noted.
“The widespread use of Oxytocin is one of the major reasons for the maternal mortality rate reducing in the country,” said Malini Aisola of AIDAN, speaking to Newsclick.
“We had argued before the court that restricting the manufacture and sale of Oxytocin to just one unit was bound to lead to a crisis in its availability, and consequently a severe public health crisis.”
As the bench of Justice S Ravindra Bhat and Justice AK Chawla noted, “the possibility or danger of concentrating production in one unit (KAPL) and the inherent vulnerability (i.e. shutdown of operations on account of unforeseen situations like fire, disasters, strike, etc) does not seem to have been weighed in at all.”
“Nor does the risk in the scarcity of the drug on account of failures or gaps in availability of Oxytocin formulation for human medical use, pan India, have been taken into account.”
The judgement spoke of the “overwhelming interest of pregnant women and new mothers, in stemming postpartum bleeding, which can be most effectively achieved by availability of Oxytocin in a dependable manner.”
The court gave primacy to the rights and the interests of women in the country by stating that while the case involved more than one public interest — “the first interest is that of the consumer public, particularly, the human consumer public and the pregnant women who are likely to be affected by any deterioration in the nature and quality of Oxytocin supplies.”
“This interest cannot be overstated,” the judgement said, noting that the “steep fall in maternal mortality” as shown by central government data was possible because of the ability to stem post-partum haemorrhage.
Moreover, the court noted, that not only was the evidence of Oxytocin misuse in the livestock industry provided by the government not convincing, but that there was also “no scientific evidence about long term adverse impact because of Oxytocin use on milch cattle – i.e. cows and buffaloes.”
As Aisola said, “The order is very well-reasoned out and strong.”
It was reported on August 3, that the Drug Technical Advisory Board had approved a proposal to amend the notification and remove the proposed ban to ensure the drug’s availability for human use. But the ministry had not made any changes to the notification.
Also Read: Budget 2018 is Bad News for India’s Already Ailing Health System
Get the latest reports & analysis with people's perspective on Protests, movements & deep analytical videos, discussions of the current affairs in your Telegram app. Subscribe to NewsClick's Telegram channel & get Real-Time updates on stories, as they get published on our website.