Oxytocin Ban: A Public Health Crisis in the Offing

The move that could lead to a public health catastrophe for Indian women is unlikely to materialise as health panel suggests a government re-think.
The move by the central government suggesting a ban on private manufacture, import and sale of oxytocin citing its misuse in the livestock industry is scheduled on September 1. However, it is now facing flak across spectrums. On the 3rd of August, The Drug Technical Advisory Board, (DTAB) chaired by S. Venkatesh, approved a proposal to amend the health Ministry notification to reconsider the proposed ban in its present form to ensure that it is available for human use.
The DTAB also recommended that the government delete the clause in the notification related to direct sales of the medicine to registered hospitals and clinics by means of the Jan Aushadhi and Affordable Medicines and Reliable Implants (AMRIT). The panel has also suggested the deletion of the clause that bans retail sales through chemists.
The ministry is yet to issue comments over the recommendations of the panel and has not revoked the notification yet.
In April this year, the Ministry of Health (MOH) had issued an initial notification suggesting the ban in light of the failure of the regulators to curb illegal sales. The enforcement of the order was set to take place from July 1, this year, but was later extended to September 1, through a notification issued in June.
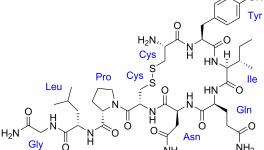
The initial announcement of the ban raised an alarm since it could have long term consequences on women’s health. The drug plays a crucial role in controlling post-birth bleeding and is used in preventing postpartum hemorrhage in women.
Oxytocin is considered essential by WHO for maternity care globally and is listed as a ‘life saving’ drug. The drug is also included in the national list of essential medicines. India has witnessed significant reduction in its maternal mortality rates. Since the brunt of the ban will be faced largely by women in India, it is likely to jeopardize this progress.
Swarnima Bhattacharya, founder of TheaCare, (Women’s health companion magazine) speaking to NewsClick on the ban stated, “Restricting its manufacture and sale will lead to prohibitive costs, which will bring in the question of affordability. The decision is not at all well thought-out, and does not take into account medical exigencies, as a result of which, as always, women will suffer because they are not priority.”
The Ministry has stated that the move is not exactly a ban as the task of producing the drug to meet the high demand nationwide is now entrusted in the Karnataka based Public Sector Undertaking- Karnataka Antibiotic Private Limited (KAPL).
However, concerns are being raised since the firm has no history of producing the drug in the past.
The question of availability of the drug with respect to the company’s monopoly over production is also critical. Currently, the drug is available in about 28 brands and 22 generics to meet the high demand and to ensure affordability. With KAPL being made the sole manufacturer of the drug, the prices are expected to soar. The medicine also comes under the DPCO 2013 ceiling price, with the maximum cap on its rate being set at rupees 17. With the implementation of the notification, a substantial jump is expected from approximately 4 rupees to about 15- 17 rupees/vial as indicated by the KAPL.
Dr. Amit Sengupta of the Jan Swasthya Abhiyan (JSA) says, “The problem is not the monopoly of the PSU, but the preparedness it has to meet the demand and the consequences of the ban in relation to women’s health.”
With the deadline of the ban approaching, KAPL is attempting to hurriedly put together multiple distribution centers across the country. The monopoly of distribution with the PSU may bring with itself some other risks during emergency conditions, such as how the supply of the medicine would be met with if the workers go on a strike or if there is an environmental calamity restricting supplies. At present, the plan to implement the ban does not offer any alternatives to accommodate such circumstances.
The ban has already spiraled into a public health crisis with doctors panicking over the availability of the drug. The 10 fold increase in the price of oxytocin has led many to panic buy stocks in advance as it is unlikely for small nursing homes to procure the drug at a price as high as the KAPL is offering.
The ban also faces a legal challenge, with the US-based manufacturer- Mylan knocking the doors of the Delhi High Court since the licenses of the company alongside other manufacturers such as Pfizer and Novartis were revoked last week.
The main reason for the ban is cited as the misuse of oxytocin in the livestock industry. Animals are injected with the drug to increase milk production. Activists campaigning for animal rights have raised concerns over the drug affecting the lifespan of cows and in some cases leading to infertility in the animal.
While the concerns related to animals require serious attention, the ban comes as an ill thought out plan reflecting a failure to envisage the repercussions it would have on women’s health across the country. Instead of stricter regulations on the use of the drug and its procurement in the animal industry, the ban becomes a policy failure for hitting the wrong note. The ban on essential life drug Oxytocin in a bid to protect Gau Mata puts at risk the lives of thousands of new mothers.
There could have been possibilities of alternatives such as a robust veterinary policy and effective treatment guidelines to curb misuse instead of putting women’s health on the line.
Many have critiqued the move for valuing cows more than the country’s women instead of taking adequate measures to protect both by resorting to knee-jerk reactions such as this notification.
Get the latest reports & analysis with people's perspective on Protests, movements & deep analytical videos, discussions of the current affairs in your Telegram app. Subscribe to NewsClick's Telegram channel & get Real-Time updates on stories, as they get published on our website.